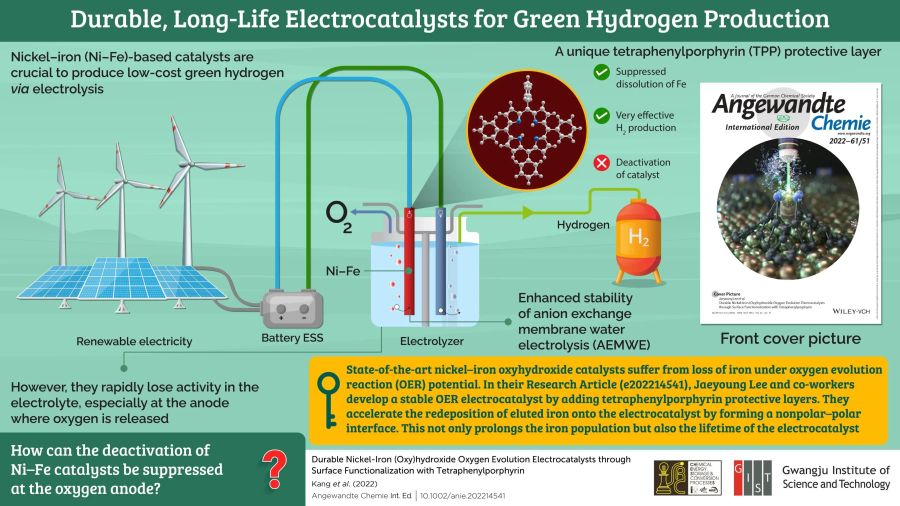
Proton exchange membrane (PEM) water electrolysis requires the use of expensive catalysts, which increases the cost of green hydrogen. Using nickel-iron (Ni-Fe) catalysts at the anode is a possible solution, but these catalysts tend to corrode and get deactivated due to the oxygen released at the anode of the water-splitting cell.
To address this issue, a team of researchers led by Professor Jaeyoung Lee of Gwangju Institute of Science and Technology (GIST) in South Korea created a protection layer for Ni-Fe catalysts that can increase their life and performance. According to Prof. Lee, “the major deactivation route for Ni-Fe catalysts is the dissolution of Fe at the oxygen emitting anode of the water-splitting cell. So, we developed an Ni–Fe-based catalyst with a tetraphenylporphyrin (TPP) protection layer. The TPP layer was able to minimize the dissolution of Fe during the oxygen evolution reaction and thus, increase the life and performance of the catalyst.”
The research team found that the TPP layer was able to shield the Ni-Fe catalyst by creating a non-polar region around the catalyst, which significantly slowed down and reduced the dissolution of the unstable Fe atoms in the catalyst. The TPP protection layer was also seen to increase the redeposition of Fe atoms back on to the catalyst. These two effects combined to increase the life of the Ni–Fe catalyst. The researchers found that the TPP protected Ni-Fe catalyst was able to sustain high hydrogen production for prolonged periods.
“Water-splitting offers a solution to meet the Paris Climate Agreement goal of being carbon neutral by 2050, since water electrolysis doesn’t emit the carbon dioxide while producing hydrogen. But the price of hydrogen production using water electrolysis is still too high. This research can help us reduce the costs by using inexpensive, non-noble materials like Ni and Fe with a protection layer. This can lead us to a more sustainable future for all of us,” said Prof. Lee.
The GIST research has been published in the Angewandte Chemie journal.