By Benoît Van Hecke, Manager, Market Development, Europe, Nickel Institute
As it gets colder, even freezing, we tend to lose control over things. Whether it’s people or vehicles, we’ve all been down that slippery slope. But how does nickel enable us to get a grip on technology when the thermometer ominously descends towards the absolute minimum?
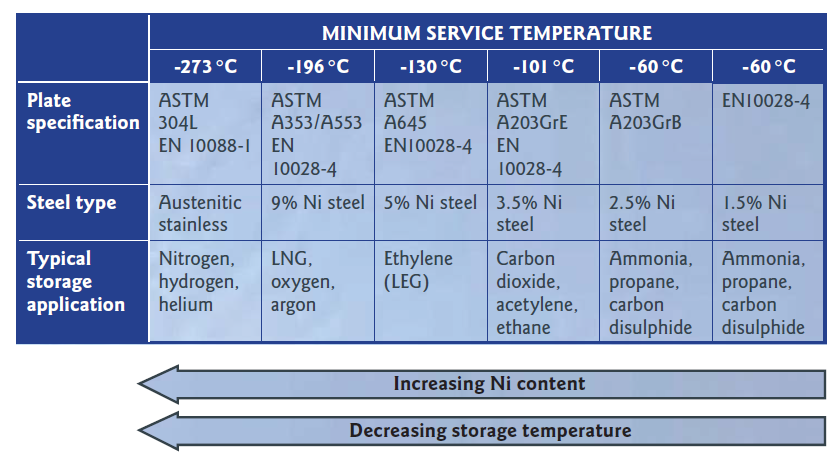
Materials for subzero temperatures
Austenitic stainless steels (typically with 7 to 25% Ni) exhibit excellent toughness at very low (cryogenic) temperatures. Toughness is the ability of a material to absorb energy without breaking, and is essential in many engineering applications. Constructional steels have good toughness at room temperature, but with decreasing temperature, the ferritic structure becomes progressively more brittle, making such steels unsuitable for service at cryogenic temperatures. In contrast, the common nickel-containing austenitic stainless steels retain good toughness even to liquid helium temperatures.
A second family of materials that exhibits good cryogenic properties is the nickel-alloyed steels. The addition of 1.50 to 9.00% Ni to a steel matrix, together with appropriate heat treatment confers the necessary toughness and strength at temperatures from -60 °C down to -200 °C.
Without chromium in the mix however, these steels are not inherently corrosion resistant, like stainless steels.
Arctic service
Arctic areas can be tricky places to recruit staff, weld metal and transport goods to and from. The most critical factor with regard to steels in arctic service is temperature. Temperatures of -50 °C or lower are no exception and are below the ductile-to-brittle transition temperature for most carbon-manganese structural steels used for construction in the Arctic oil fields.
Other factors determining material selection for arctic service in the oil and gas industry are corrosion resistance and weldability. The former is not as important for structural applications as for the production and transportation of corrosive oil and gas. It is important for the welds to retain the enhanced fracture toughness of the base metal used for arctic service. If the weld has inferior toughness, fracture will occur regardless of the quality of the base metal. Another example comes from scientific infrastructure on Antarctica, where a Belgian research station was erected in 2008. The façade is made of 1.50 mm 304L (UNS S30403) stainless steel panels. This material was chosen because austenic stainless steel (304L) continues to exhibit sufficient strength and toughness well below even -60 °C.
LNG
As energy demand constantly grows and energy transportation and storage become more and more crucial, liquefied natural gas (LNG) looks attractive, because of its ratio of volume to energetic density. The volume of natural gas in its liquid state is about 600 times smaller than its volume in its gaseous state in a natural gas pipeline. The liquefaction process makes it possible to transport natural gas to places where there are no natural gas pipelines and to even use natural gas as a transportation fuel.
None of this would be possible without nickel, since natural gas only becomes liquid below -163 °C. As previously described, austentic stainless steel and nickel alloyed steels are the best solutions for assembling storage and transportation vessels and for handling liquefied natural gas at such low temperatures.
Tanker design
LNG tankers predominantly sail from Qatar and Australia to areas with high energy demand such as Japan, China and South Korea. Modern tanker designs make optimum use of hull space by containing the liquefied gas in a double metallic membrane, backed by insulation. Waffled (to allow for expansion and contraction) thin gauge 304L stainless steel or thinner and flat Alloy 36 (K93600), a low thermal expansion 36% Ni alloy, are exclusively used for these cargo containment systems.

More liquefied gases Sliding further down the temperature scale from LNG at -163 °C are:
• -183 °C: liquid oxygen
• -196 °C: liquid nitrogen
• -253 °C: liquid hydrogen
• -269 °C: liquid helium
Applications are often found in industry, which relies on these products in regasified form. Hydrogen is already frequently used as an industrial precursor to ammonia in the fertiliser industry. It also holds a lot of potential to decarbonise the energy mix. Provided that sufficient hydrogen can be produced (preferably green hydrogen from wind or solar power), burning it as a fuel produces just water and no carbon dioxide. The steel industry being energy intensive, several producers are already looking at reducing their carbon footprint by increased use of hydrogen.
There would be no space exploration without liquid gases. Hydrogen is a light and very powerful rocket fuel. Combined with an oxidiser such as liquid oxygen, liquid hydrogen proves the most efficient of all rocket propellants.
Finally, what could possibly cool a superconducting magnet to sustain a plasma state? The answer is simple: liquid helium. But the temperature isn’t. At -269°C or 4 Kelvin, this is where nickel unlocks its full potential…

About the Nickel Institute
The Nickel Institute is the global association of leading primary nickel producers. Its mission is to promote and support the proper use of nickel in appropriate applications.
For information, visit: www.nickelinstitute.org






